Get 5 free video unlocks on our app with code GOMOBILE
Snapsolve any problem by taking a picture.
Try it in the Numerade app?
Solved step-by-step
What is the change in the oxidation number of manganese in the
following?
KMnO4 → MnSO4
Increase by four units.
Increase by three units.
Decrease by four units.
Decrease by five units.
Solved by verified expert
This problem has been solved!
Try Numerade free for 7 days
00:52
In the oxidation reaction of toluene with potassium permanganate in basic conditions, manganese dioxide is produced. What is the change in the oxidation state of manganese?+7to +2+7to +41 to +4+6 to +4
01:11
In the following redox reaction, the oxidation number of manganese changes fromMnO4¯(aq) + 2H2O(l) + 3Co2+(aq) —-> MnO2(s) + 3Co3+(aq) +4OH¯(aq)
03:15
Calculate the oxidation state of manganese in each of the following: Atom/Molecule/Ion Oxidation State of Manganese Mn Mn2+ MnO2 MnO4- MnO42- MnCl2
02:24
When MnO4- reacts to form Mn2+, the manganese in MnO4- is reduced as its oxidation number decreases.
01:12
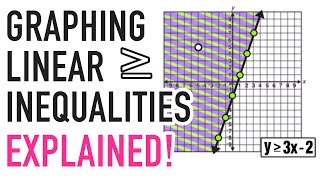
What is the oxidation number of manganese in $\mathrm{KMnO}_{4} ?$
Transcript
Hello, let’s have a look on the question here. We have given changes, k 42 and minus 24. Potassium permangnetthat is camineroxidation number of potassium plus 1. Oxidation number of oxygen, that is o, is equal to minus 2 point. Let oxidation stay of m n. Magnese is x, sum of oxidation state of tons of k, m and 4 is equal to 0, so 1 milty oxidation number of potassium plus 1, that will have an oxidation number of magnese plus 4 multiply by oxidation number of oxygen is equal to 01 tegamus 1 Plus 1 digamex plus 4 by the p minus 2 point that is equal to 01 plus x. Minus 8, that…
Enter your parent or guardian’s email address:
Already have an account? Log in
Create an account to get free access
or
PASSWORD